Question 15 1 Point Liv is trying to decide which molecules to use for her chemistry reaction. Question 14 1 Point Calculate the average atomic mass for the following isotopes: Hydrogen: hydrogen-1 (1.0078 amu) at 99.989%, hydrogen-2 (2.0141 amu) at 0.0115% Question 13 1 Point Calculate the average atomic mass for the following isotopes: Boron: boron-10 (10.0129 amu) at 19.9%, boron-11 (11.0093 amu) at 80.1%. Question 12 1 Point Identify the element that fits into each of these categories on the periodic table: Alkaline Earth Metals 
Question 11 1 Point Identify the element that fits into each of these categories on the periodic table: Noble gases Question 10 1 Point Identify the element that fits into each of these categories on the periodic table: Nonmetal Question 9 1 Point Which ion has +3 charge, 28 electrons, and an atomic mass of 70 amu? Question 8 1 Point How many electrons do you need to add to the orbit to make a stable fluorine atom? Question 7 1 Point How many electrons do you need to add to the orbit to make a stable Beryllium atom? Which element does this describe? Is it a neutral atom, isotope, or an ion? What is the mass number? What is the net charge? Question 6 1 Point An element has 6 neutrons, 4 electrons and 4 protons. 
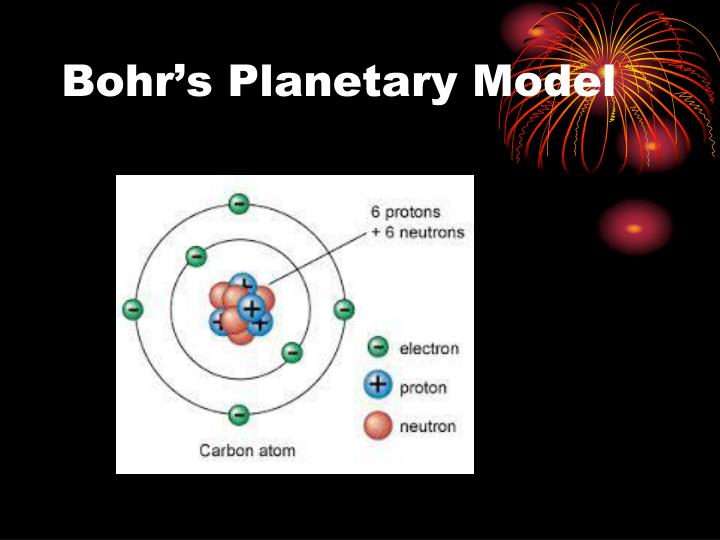
Question 5 1 Point An element has 4 neutrons, 3 electrons and 4 protons. Describe the 5 main differences between the Bohr Atom Model and the Quantum Mechanical model. Question 4 1 Point You have been tasked to give a report about the differences in the Bohr Model v. Question 3 1 Point How does an orbit differ from an orbital? Question 2 1 Point Describe the difference between the principal quantum number and the angular momentum quantum number. 
123–150.Why is it necessary for the spins of two electrons that occupy one orbital, to be in the opposite directions? What is that law called? (eds.) Science after the Practice Turn in the Philosophy, History, and Social Studies of Science, pp. In: Soler, L., Zwart, S., Lynch, M., Israel-Jost, V. Woody, A.: Chemistry’s periodic law: rethinking representation and explanation after the turn to practice. Weber, E., Lefevere, M.: Unification, the answer to resemblance questions. Harvard University Press, Cambridge (2008) Scerri, E.: Causation, electronic configurations and the periodic table. Scerri, E.: The Periodic Table: its Story and its Significance. Ross, L.: Causal explanation and the periodic table. Moseley, H.: The high-frequency spectra of the elements Part II. Moseley, H.: The high-frequency spectra of the elements. Zhurnal Russkeo Fiziko-Khimicheskoe Obshchestv 1, 60–77 (1869) Mendeleev, D.: Sootnoshenie Svoistv s Atomnym Vesom Elementov. European Journal for Philosophy of Science 4, 337–359 (2014) Kuhlmann, M., Glennan, S.: On the relation between quantum mechanical and neo-mechanistic ontologies and explanatory strategies. (eds.): The Routledge Handbook of Mechanisms and Mechanical Philosophy. Glennan, S., Illari, P.: Introduction: Mechanisms and Mechanical Philosophies, in Glennan, S., Illari, P. Glennan, S.: The New Mechanical Philosophy. Brush, S.: The reception of Mendeleev’s periodic law in America and Britain. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
